Articles from mBIOTA Labs

mBIOTA Labs, an emerging medical food company revolutionizing medical nutrition for the management of gastrointestinal (GI) conditions, announced today that two abstracts featuring mBIOTA Elemental™ Diet are being presented at Digestive Disease Week 2026 in Chicago, Illinois. These abstracts demonstrate the clear effectiveness of mBIOTA Elemental as a therapeutic tool for patients living with Irritable Bowel Syndrome (IBS) and explore the mechanism by which the mBIOTA elemental diet modulates the microbiome in overgrowth conditions.
By mBIOTA Labs · Via Business Wire · May 4, 2026

mBIOTA Labs, an emerging medical food company revolutionizing medical nutrition for the management of gastrointestinal (GI) conditions, announced that four clinical studies leveraging mBIOTA Elemental™ Diet and two studies using mBIOTA Labs’ 2025 national gut health survey were presented at the American College of Gastroenterology (ACG) 2025 Annual Scientific Meeting in Phoenix, Arizona.
By mBIOTA Labs · Via Business Wire · November 5, 2025

mBIOTA Labs, an emerging medical food company revolutionizing medical nutrition for the management of gastrointestinal (GI) conditions, announces that their flagship product, mBIOTA Elemental™ Diet, has shown effectiveness at managing the gut microbiome beyond just gut bacteria. The first clinically-proven palatable elemental diet, initially studied as a solution for small intestinal bacterial overgrowth (SIBO) and intestinal methanogen overgrowth (IMO) as focused on reducing intestinal bacteria and related symptoms of such gastrointestinal disorders, has now been demonstrated to effectively reduce Methanobrevibacter smithii (M. smithii), an abundant methane-producing archaea in humans that is known to cause IMO.
By mBIOTA Labs · Via Business Wire · May 5, 2025

mBIOTA Labs, an emerging medical food company revolutionizing medical nutrition for the management of gastrointestinal (GI) conditions, highlights expanded data findings from its prospective clinical trial indicating that its flagship product, mBIOTA Elemental™ diet, is a highly effective solution for the management of gastrointestinal disorders, specifically Small Intestinal Bacterial Overgrowth (SIBO) and Intestinal Methanogen Overgrowth (IMO). The clinical research findings are published in Clinical Gastroenterology and Hepatology, awarded by the 2024 American College of Gastroenterology Annual Meeting, and were presented at the 2024 Digestive Disease Week and internationally at the 2024 Federation of Neurogastroenterology and Motility Meeting.
By mBIOTA Labs · Via Business Wire · April 7, 2025
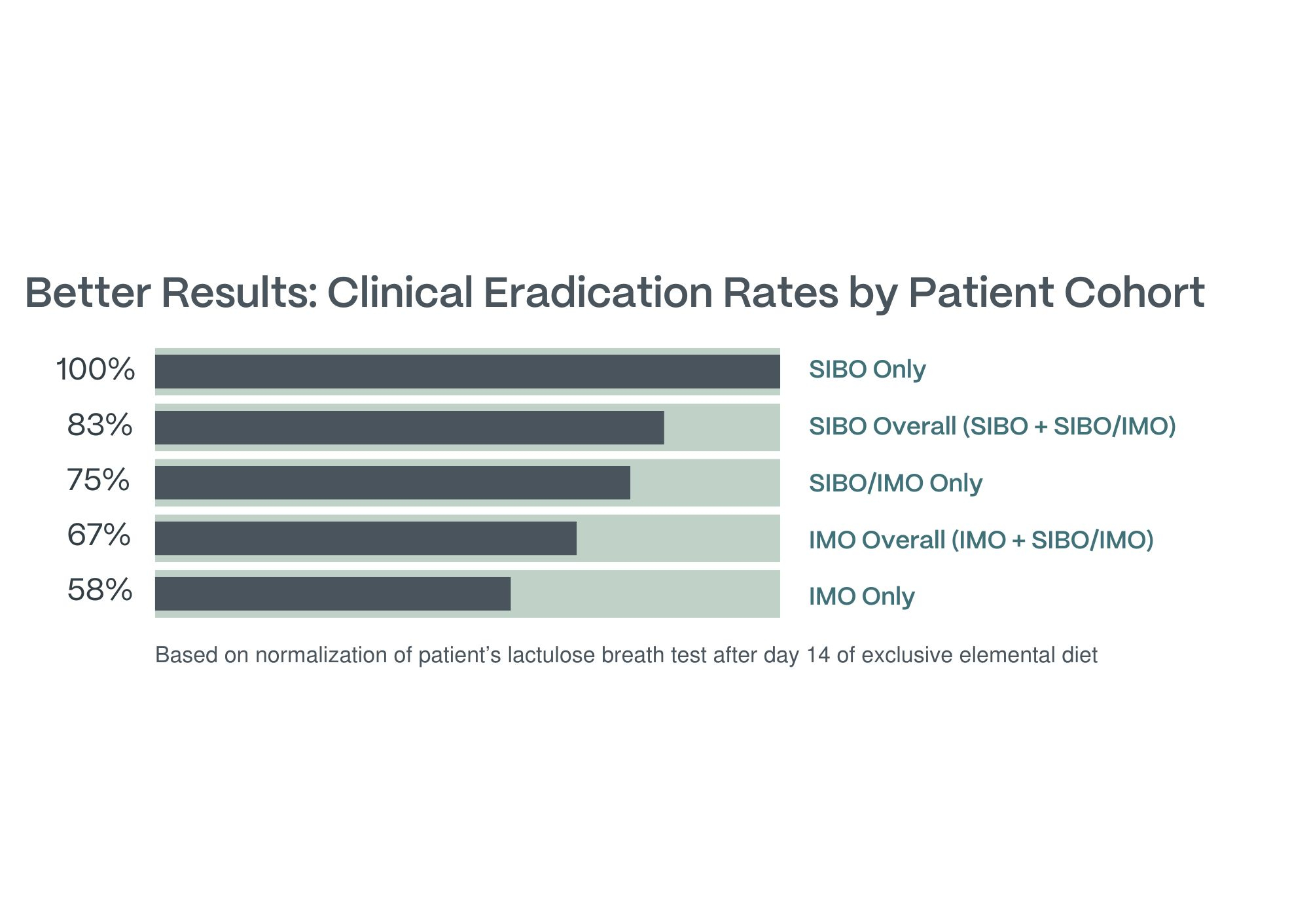
mBIOTA Labs, a medical food company revolutionizing oral nutrition through their proprietary palatable elemental diet, mBIOTA Elemental™, today announced the initial study results of their first prospective clinical trial assessing the efficacy of the mBIOTA Elemental™ diet in patients with small intestinal bacterial overgrowth (SIBO), intestinal methanogen overgrowth (IMO), or a combination of the two.
By mBIOTA Labs · Via Business Wire · May 20, 2024
